3D Printing In Healthcare: Where Are We In 2021? (Updated)

3D printing is creating an enormous opportunity for the medical industry. According to a report by market research firm, SmarTech Analysis, the market for medical 3D printing is currently estimated to be worth $1.25 billion. By 2027, the market value is set to grow to $6.08 billion. Clearly, the potential for 3D printing within healthcare is huge. Multiple sectors within the medical industry are benefiting from 3D printing, including orthopaedics and dental. The technology is offering exciting new ways to provide personalised care and create better-performing medical devices. In today’s article, we’ll be exploring the key uses of 3D printing in medicine and how they will evolve, the challenges to wider adoption and what the future looks like for medical 3D printing.
3 key applications of 3D printing in healthcare
1. 3D-printed orthopaedic implants

Orthopaedic implants — medical devices used to surgically replace a missing joint or bone — are one of the applications that benefit the most from 3D printing. The technology enables medical professionals to create better-fitting, longer-lasting and higher-performing implants. The first use of 3D printing for orthopaedic implants dates back over a decade, with the first 3D-printed implants manufactured around 2007. In 2010, Adler Ortho Group, an early adopter of Arcam’s Electron Beam Melting (EBM) metal 3D printing technology, received the first FDA approvals for implants created by 3D printing.Today, the technology can be used to make a wide range of implants, including spinal, hip, knee and skull implants. By the end of 2019, it’s estimated that over 600,000 implants will have been produced with 3D printing. By 2027, this number could increase to four million. In addition to EBM, Selective Laser Melting is another metal 3D printing technology used by orthopaedic manufacturers. Both technologies are optimised to work with biocompatible metals like titanium, and can produce many complex implants in one batch. For example, US-based manufacturing company, Slice Mfg. Studios, says that each of its Arcam Q10 EBM machines can produce around 70 acetabular hip cups every five days. One of the factors driving the demand for 3D-printed implants is the potential for enhanced implant performance. Thanks to the design flexibility offered by 3D printing, implants can be designed with porous surface structures, facilitating faster integration between a living bone and the artificial implant. The orthopaedic medical device industry is dominated by a small number of multi-billion dollar medical companies, notably Stryker, DePuy Synthes, Medtronic and Smith & Nephew, all of whom are actively exploring AM for a range of innovative medical devices. For example, Stryker has recently launched 3D-printed implants, including the 3D-printed Tritanium TL Curved Posterior Lumbar Cage. This hollow-bodied spinal implant received FDA approval in March 2018. 3D printing in the orthopaedic sector is largely being used to enhance the design of standard implants to improve their performance. The biggest potential, however, lies in the production of patient-specific implants, which remains largely untapped due to regulatory issues that we’ll be discussing below. Despite current challenges, 3D printing individualised implants represents a key opportunity for the orthopaedic segment, and one that will see tremendous growth in the years ahead.
2. Personalised surgery
3D printing technologies are increasingly being used to develop patient-specific models of organs and surgical tools, using patient’s own medical imaging.
Patient-specific anatomical models
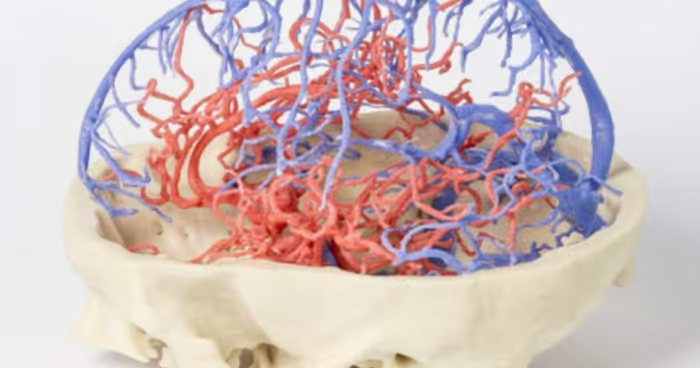
Anatomical models are currently one of the most widely adopted applications of 3D printing in the medical industry. The accessibility of medical CAD/CAM software and lower-cost desktop 3D printers is increasing, enabling more hospitals to establish 3D printing labs. In such labs, medical professionals can produce high-accuracy 3D-printed models to assist in presurgical planning. 3D-printed anatomical models help surgeons evaluate better treatment decisions and plan their surgeries more accurately. The process begins by taking CT or MRI scans. The scans are then analysed and modified to remove undesired areas and keep the regions of interest (a process known as segmentation). Bones, vessels and solid organs all need to be modelled in different ways. Once the digital model is created, it is converted into an STL file format, prepared for printing and sent to a 3D printer. Rady Children's Hospital in the US, for example, has established a 3D Innovations Lab, where medical engineers 3D print dozens of models per week. “We actually sit and analyse the models and that helps us understand what is the optimal approach to repairing the defect,” said John Nigro, M.D., chief of Rady Children’s cardiac surgery, speaking in an interview with KPBS News. By preparing for surgery using a 3D-printed model, surgeons can reduce the time a patient spends in the operating room. Ultimately, this leads to fewer complications and a better long-term outcome for the patient.Beyond surgery, 3D printers that can replicate patient organs are excellent tools for medical research, education and training. For example, holding a model and viewing the pathology from different angles helps students understand the steps involved in surgery more clearly.
Enhanced surgical tools

Another area where 3D printing is making an impact is personalised surgical tools. Surgical instruments, like forceps, hemostats, scalpel handles and clamps can be produced using 3D printers. Creating personalised surgical instruments offers many benefits. They facilitate faster and less traumatic procedures, increase a surgeon's dexterity and support better surgery outcomes. For such applications, 3D printing companies have developed biocompatible materials that can withstand sterilisation, including high-performance thermoplastics like Ultem, PEEK, nylon and also metals like stainless steel, nickel and titanium alloys.German medical device company, endocon GmbH, has used metal 3D printing and stainless steel alloy (17-4 PH) to develop a surgical tool for hip cup removal. The goal was to make the process of removing hip cups easier and faster. Typically, the procedure lasts around 30 minutes and is carried out with a chisel, which risks damaging bone and tissue. This, in turn, can make the surface of a bone uneven and the insertion of a new implant even more difficult.The new tool features additively manufactured blades, which enable more precise cutting along the edge of the acetabular cup, allowing surgeons to remove the cup within three minutes. In terms of biocompatibility, the 3D-printed blades are said to lead to a more consistent outcome of hip cup replacement, with the rejection rate reduced from 30% to less than 3%. Furthermore, the production and finishing of 3D-printed blades take just three weeks, while the costs are reduced by 45%.As advantages of 3D printing for surgical devices become more widely recognised, stories like this will become much more common in the future.
3. Medical & Dental devices
Medical and dental devices like prosthetics, braces, dentures, restorations and clear aligners can significantly benefit from 3D printing. According to a report, the global 3D printing medical devices market was valued at approximately $890 million in 2017. The market is expected to generate around $2.34 billion of revenue by the end of 2024, pointing to a significant growth opportunity. Low-cost personalisation is a key benefit, driving the adoption of 3D printing for medical & dental devices. A 3D printer requires only a digital file to produce a device, which makes it possible to customise a design more easily and produce many different devices in one batch. With traditional manufacturing like moulding, each customised device would require special tooling, making customised production economically unviable. With 3D printing, prosthetic limbs are becoming much more affordable and faster to produce. Furthermore, the technology can be used to create prosthetics tailored to the patient’s anatomy, thereby improving the fit of the prosthetic. Increasingly, 3D printing is being used to create prostheses for children. Children’s rapid growth means that they can quickly outgrow traditional prostheses. As a result, it’s necessary to replace them with a larger size version every couple of years. The lower costs associated with 3D printing make this a far better-suited manufacturing option.Nonprofit Limbitless Solutions, for example, provides children with an opportunity to customise their prosthetic limbs by choosing from a collection of colour palettes and designs, which reflect their personality. The designs are then 3D-printed using FDM technology from Stratasys and durable plastics like ABS. In addition to the ability to create complex prosthetic designs, 3D printing reduces the cost of production. In some cases, Limbitless’ prosthetics cost 40 times less than a traditional prosthetic limb.
Dental 3D printing
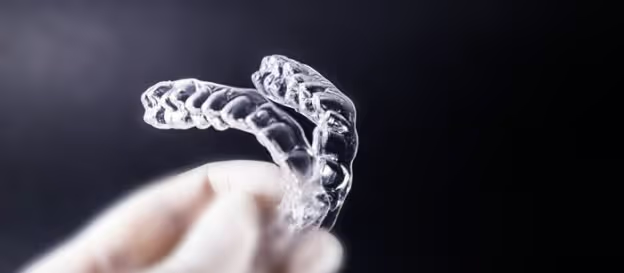
3D printing is predicted to make a huge impact in the dental sector. A report by SmarTech Analysis suggests that revenues for 3D-printed dentistry will grow to $3.7 billion by 2021, and the technology will become the leading production method for dental restorations and devices worldwide by 2027. “We’re seeing that 3D printing is becoming one of the key tools in areas like dental care and dental restoration. The digital thread there has been largely developed all the way from intraoral scanning to the workflows and the planning — not just in the lab, but also in the dental clinic. So there you can see a market that is ready for mass adoption, ” says Avi Reichental, Founder of XponentialWorks, speaking in an interview with AMFG. Clear aligners — invisible teeth straightening devices — are perhaps the biggest use case of 3D printing in dental today. Major clear aligner companies like Align Technology and NextDent use 3D printing to create hundreds of thousands of moulds for clear aligners. Over the next five years, 3D printing is predicted to evolve to the point where it can be used to create clear aligners directly.
3D printing in healthcare: regulatory perspectives
However, unlocking the full potential of 3D printing for healthcare is not without challenges. Currently, the lack of a comprehensive regulatory framework for 3D-printed medical and dental products is one of the industry’s biggest barriers. Several regulatory bodies are working on developing standards for 3D printing in healthcare. The U.S. Food and Drug Administration (FDA), for example, issued a guidance “Technical Considerations for Additive Manufactured Devices” in December 2017. The guidance highlights the technical considerations and recommendations for design, manufacturing and testing of 3D-printed medical devices.
Standardising medical devices

There are three major classes of medical devices, based on the level of harm they may pose to a patient.
- Class I: Devices represent a low risk to the patient. Examples include nasal oxygen cannulas, manual stethoscopes and hand splints.
- Class II: These devices are more invasive and represent a moderate risk to the patient.The majority of medical devices are class II, with examples including tracheal tubes, bone plates and elbow joint radial prostheses.
- Class III: These are devices that represent the highest risk to the patient and include aortic valves, constrained metal hip prostheses and coronary stents.
While a 3D-printed prosthetic may be classified as Class I, or low risk, the technology has progressed to enable more advanced implants and tools to be produced that encompass Class III, or high risk, medical devices.To certify devices under Class I, manufacturers have to prove that the final product is largely the same as a device that is already on the market.For Class II and III medical devices, FDA and other regulatory bodies have yet to issue guidelines on pre-market approval of 3D-printed medical devices. These are innovative products, and would likely require modifications to an existing medical regulatory framework.To date, more than 100 medical devices have been approved by the FDA, most of which fall under Class I medical devices.Patient-specific devices are the most complicated case to regulate. Traditionally manufactured medical devices are standard, one-size-fits-all. However, with a product that is customised, it can be challenging to test each and every single device that is custom-made.Going forward, to create more opportunities for personalisation, regulatory bodies should find ways of how to pre-approve custom devices. Currently, it's challenging because the requirements for approval are developed to certify off the shelf implants and instruments. Therefore, regulatory bodies need to focus on how they can address the differences between people rather than only similarities to enable this level of personalised care. Organisations like the FDA are trying to overcome this issue by setting maximum and minimum sizes or features for custom-created devices.
Reimbursement challenges
Lack of reimbursement can be a major barrier for hospitals thinking about establishing a 3D printing lab. Within the healthcare industry, reimbursement describes the payment that a hospital, doctor, diagnostic facility or other healthcare providers receive for providing a medical service. Often, a health insurer or a government payer covers the cost of all or part of a provided care.While an FDA-approved 3D-printed joint implant or bone fixator may be reimbursed, 3D models of a patient's anatomy and professional fees often are not.Fortunately, healthcare organisations are actively working to change that. For example, the American Medical Association (AMA) has recently approved four Category III Current Procedural Terminology (CPT) codes for 3D-printed anatomical models and personalised 3D-printed cutting or drilling tools. The four new codes will allow radiologists and other clinicians to seek reimbursement for 3D printing services. Another driver for using CPT codes is to ensure all of the production steps of a 3D-printed medical device are recorded. The data collected through the codes will be used to support the FDA approval processes. For medical 3D printing, the introduction of the codes represents a key milestone on the way towards widespread adoption of 3D printing in healthcare.
The future of 3D printing in healthcare
3D printing is set to hold a fundamental position in the future of healthcare. Today, the technology is facilitating surgical teams both inside (surgical tools) and outside (anatomical models) the operating room. Furthermore, it creates dental products cheaper and faster and enables personalised care through custom-produced instruments and implants. In 2019, leading hospitals and labs are adopting 3D printing as part of their medical practices and research efforts. This serves as yet another validation of the technology’s value for medical applications.Supporting the technology’s evolution within the medical industry is the collaborative effort to create a single, cohesive set of standards and test methods for 3D-printed medical products. Overcoming current regulatory and legal challenges will certainly help to guide the technology going forward.Looking beyond the applications discussed in this article, the potential of 3D printing is expanding to other healthcare sectors. Among them are bioprinting and regenerative medicine, ophthalmology and pharmaceuticals. Here, 3D printing is still very much in its early stages, but its potential is significant. All in all, the future of healthcare will look very different from the healthcare of today — and 3D printing will be one of the key technologies to drive forward this exciting and meaningful transformation.

.svg)
.svg)






.avif)

.svg)

