How Digitisation Is Key to the Future of Medical 3D printing

2020 was a pivotal year for the healthcare industry. As hospitals were flooded with COVID-19 cases, the demand for healthcare equipment and protective devices surged, leaving medical supply chains disrupted.
With the supply chain shortages, many hospitals turned to local manufacturers to help produce components for ventilators, face shields, respiratory masks and testing swabs.
Many of these local companies were 3D printing service bureaus. They provided an opportunity to design, evaluate and manufacture parts in a matter of days, so hospitals could receive vital parts as soon as possible.
In a sense, the onset of the pandemic threw a spotlight on 3D printing and its potential for fast, flexible production of critically needed parts.
However, the medical industry recognised this potential long before the pandemic. Hospitals and medical device manufacturers alike have been using 3D printing for the ever-growing range of applications for years.
From anatomical models and surgical instruments to implants and prostheses, the opportunities for medical 3D printing are truly vast.
3D printing – with its capacity for personalisation and flexible production – is set to hold a fundamental position in the future of healthcare. The medical AM market is already estimated at more than $1 billion, and this number is only set to increase, reaching $6 billion by 2027.
But to make full use of AM in medical, companies adopting the technology need to surmount a few challenges in managing data, complying with regulations and ensuring cost-effective, repeatable AM production.
Below, we take a closer look at each of these challenges and the potential of digitisation and software for solving them.
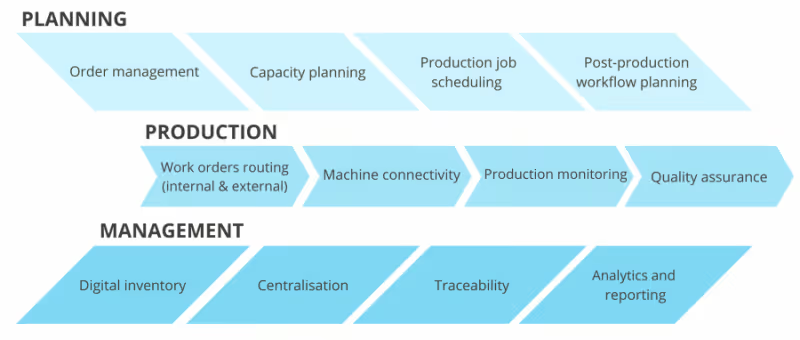
Managing data in medical additive manufacturing
Accurately collecting and maintaining complete data about how a medical device was manufactured and inspected is paramount. However, data collection in AM remains a highly manual process, ripe for errors and audit data gaps.
When one system is tracking the progress on projects, while the other is used for production scheduling, and the third one – for quality control, the information about the whole process becomes extremely fragmented.
Essentially, each system is managing its version of the truth, and synchronising data across systems eats time that could be used more efficiently.
A better solution for improved data handling would be to digitise and centralise production management processes. Digitisation makes it easier to connect and integrate siloed systems, while centralisation helps to keep essential production data in one place, update it instantaneously if any changes occur and track these changes.
The best way to digitise and centralise additive manufacturing production data is arguably through a manufacturing execution system (MES).
The capabilities of modern MES software in ensuring data transparency are truly vast. Essentially, additive MES solutions can help establish a digital workflow, which allows you to govern the ability to capture, enrich and share well-defined, accurate, coherent and up-to-date information across the enterprise and beyond.
For example, each part receives a unique serial number in MES, with which you can track each step a part went through, for example, post-processing operations and inspection procedures.
Importantly, you can integrate an MES with your AM systems to monitor 3D printers in real time and collect data about their performance.
The ability to store relevant production parameters, such as job print time, temperature, laser intensity etc., as well as print results, gives the production team a clear view of which parameters resulted in the highest-quality printing jobs or which led to a failure. Analysing this data can give a boost to quality, ultimately driving productivity improvements.
Ensuring repeatable production
While AM technology matures fairly quickly, production repeatability can still be difficult to achieve. Various factors may cause repeatability issues, including material quality, errors during production scheduling, part orientation within the build platform, and machine calibration.
In the manufacturing of medical devices, it’s critical that all production steps and parameters are determined, tracked, and controlled to allow for consistency between parts. This requires a well-designed and executed manufacturing process. One that will be challenging to establish when production processes are isolated and don’t work in sync.
When experiencing a lack of production repeatability, a solution is to enforce repeatable additive manufacturing through workflow automation.
Workflow automation makes it possible to establish rules where tasks are automatically triggered and routed between people, technology and data.
So, for example, when you have an order to produce a titanium part in a Z-axis, the part can be assigned to a specific machine according to defined parameters automatically, reducing the chance of scheduling a part in the wrong orientation or to a wrong 3D printer.
Workflow automation is an essential part of modern additive MES solutions. On the way to scale, MES helps you identify, replicate and enforce what works, as well as controlling and revising what does not. In this way, it eliminates inconsistencies arising from manual, paper-based processes, strengthening your ability to 3D print medical parts repeatedly.
Discover more about AM automation: Where Are You in Your Additive Manufacturing Automation Journey?
Meeting regulatory and quality requirements for 3D-printed medical devices
As with any product used in the medical industry, manufacturers of 3D-printed medical devices must comply with standards and regulations provided by healthcare agencies like the FDA and the EMA.
In AM, rather than a focus on compliance, regulators are looking for manufacturers to shift their focus to ensuring quality is built into products and processes rather than inspected out at the end.
Doing so without a specialised digital system to support control and monitoring of production processes is nearly impossible.
Modern additive MES can help companies validate processes, remove a lot of the pain associated with audits, and reduce both cost and risk in quality control.
Let’s take the auditing process as an example. If the auditing team comes to a 3D printing facility and demands documented evidence that specific AM processes are in control, the traditional paper-based systems for storing data make it difficult to quickly provide the required information.
The historic data may well be stored offsite and could take days to retrieve. The more time the inspector must spend at the facility, the more information thet may request, increasing the workload for staff supporting the audit.
The MES controls processes and enforces procedures, and data is automatically logged with complete traceability throughout the production line. This means that even at the most basic level, having documentation and information about processes quickly available wherever and whenever needed reduces the man-hours needed to go through an audit.
As well as quickly demonstrating processes are in or back under control, the 3D printing management system further gives the regulatory body a level of comfort that they will remain this way.
Through this tight process control, reduced production variability, traceability, and secure recording of all information relating to AM production, the additive MES provides solid support for compliance to medical regulations, including ISO 13485, 21 CFR Part 11 and 820.
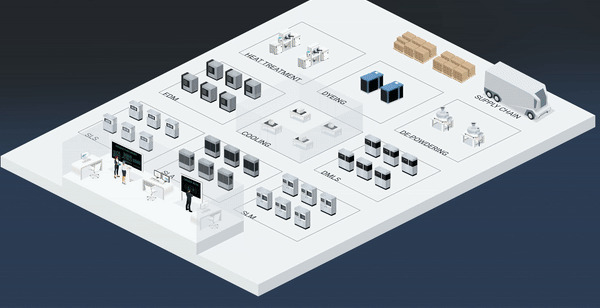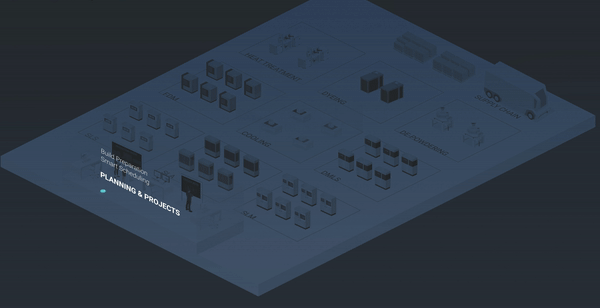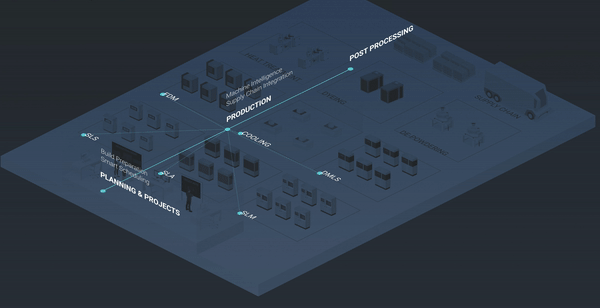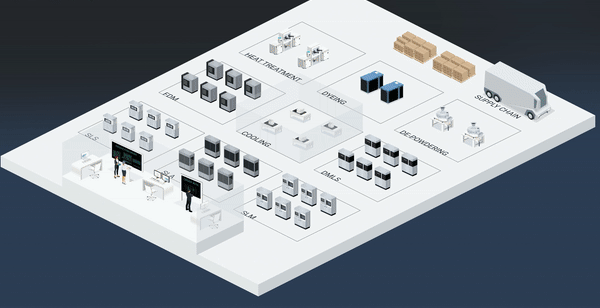
Enable the digital thread in medical additive manufacturing
Of course, the benefits of a modern, future-ready additive MES go far beyond helping to deal efficiently with regulatory requirements. As we’ve seen, there are advantages to be gained in streamlined, standardised operations, and facilitation of paperless, data-driven production management.
But the biggest benefit of an additive MES is perhaps the ability to orchestrate and connect your additive manufacturing operations into one digital thread, acting as a catalyst for continuous improvement and scalable growth.
Learn how AMFG can help you start collecting data, standardise and automate processes with MES today

.svg)
.svg)





.avif)


.svg)

